ILSI entities around the world publish scientific articles on original research, literature reviews and gap analyses, as well as meeting proceedings in peer-reviewed journals and publications. Not one of the 1,000+ articles that ILSI has published over the last 45 years has ever been retracted. ILSI also publishes books, monographs, white papers, other scientific reports, annual reports and newsletters.
ILSI's flawless scientific publication track record, its commitment to the highest scientific standards and its adherence to rigorous scientific principles demonstrate its scientific integrity.
ILSI's publications are listed below by publication date, from the newest article to the oldest. You can also filter the list by title or publication type.
Publication Date
All Publications
15 anos da lei de biossegurança: o caso da soja GM no Brasil
2023
- ILSI Brasil
Bioactive compounds intake in the Brazilian population: Trends and determinants of socioeconomic inequalities between 2008 and 2018
PLOS ONE, 2023
- ILSI Brasil
Effect of Non-Nutritive Sweeteners on Insulin Regulation, Glycemic Response, Appetite and Weight Management: A Systematic Review
Nutrition and Food Science, 2023
- ILSI-India
This is a systematic review on the effect of non-nutritive sweeteners on appetite, weight, glycemic regulation, and gut microbiome.
Small intestine vs. colon ecology and physiology: Why it matters in probiotic administration
Cell Reports Medicine, 2023
- ILSI Europe
We provide a detailed review unfolding how the physiological and anatomical differences between the small and large intestine affect gut microbiota composition, function, and plasticity. This information is key to understanding how gut microbiota manipulation, including probiotic administration, may strain-dependently transform host-microbe interactions at defined locations.
Contribuição do eucalipto geneticamente modificado em uma perspectiva de demanda global e produção sustentável
2023
- ILSI Brasil
WP_Query Object
(
[query] => Array
(
[post_type] => publication
[posts_per_page] => 5
[type] =>
[area] =>
[committee] =>
[authors] =>
[showtitle] =>
[meta_query] => Array
(
[relation] => AND
)
[tax_query] => Array
(
)
[paged] => 4
[meta_key] => _ilsi_date
[orderby] => meta_value
[order] => DESC
)
[query_vars] => Array
(
[post_type] => publication
[posts_per_page] => 5
[type] =>
[area] =>
[committee] =>
[authors] =>
[showtitle] =>
[meta_query] => Array
(
[relation] => AND
)
[tax_query] => Array
(
)
[paged] => 4
[meta_key] => _ilsi_date
[orderby] => meta_value
[order] => DESC
[error] =>
[m] =>
[p] => 0
[post_parent] =>
[subpost] =>
[subpost_id] =>
[attachment] =>
[attachment_id] => 0
[name] =>
[pagename] =>
[page_id] => 0
[second] =>
[minute] =>
[hour] =>
[day] => 0
[monthnum] => 0
[year] => 0
[w] => 0
[category_name] =>
[tag] =>
[cat] =>
[tag_id] =>
[author] =>
[author_name] =>
[feed] =>
[tb] =>
[meta_value] =>
[preview] =>
[s] =>
[sentence] =>
[title] =>
[fields] =>
[menu_order] =>
=>
[category__in] => Array
(
)
[category__not_in] => Array
(
)
[category__and] => Array
(
)
[post__in] => Array
(
)
[post__not_in] => Array
(
)
[post_name__in] => Array
(
)
[tag__in] => Array
(
)
[tag__not_in] => Array
(
)
[tag__and] => Array
(
)
[tag_slug__in] => Array
(
)
[tag_slug__and] => Array
(
)
[post_parent__in] => Array
(
)
[post_parent__not_in] => Array
(
)
[author__in] => Array
(
)
[author__not_in] => Array
(
)
[search_columns] => Array
(
)
[ignore_sticky_posts] =>
[suppress_filters] =>
[cache_results] => 1
[update_post_term_cache] => 1
[update_menu_item_cache] =>
[lazy_load_term_meta] => 1
[update_post_meta_cache] => 1
[nopaging] =>
[comments_per_page] => 50
[no_found_rows] =>
)
[tax_query] => WP_Tax_Query Object
(
[queries] => Array
(
)
[relation] => AND
[table_aliases:protected] => Array
(
)
[queried_terms] => Array
(
)
[primary_table] => wp_posts
[primary_id_column] => ID
)
[meta_query] => WP_Meta_Query Object
(
[queries] => Array
(
[0] => Array
(
[key] => _ilsi_date
)
[relation] => OR
)
[relation] => AND
[meta_table] => wp_postmeta
[meta_id_column] => post_id
[primary_table] => wp_posts
[primary_id_column] => ID
[table_aliases:protected] => Array
(
[0] => wp_postmeta
)
[clauses:protected] => Array
(
[wp_postmeta] => Array
(
[key] => _ilsi_date
[compare] => =
[compare_key] => =
[alias] => wp_postmeta
[cast] => CHAR
)
)
[has_or_relation:protected] =>
)
[date_query] =>
[request] =>
SELECT SQL_CALC_FOUND_ROWS wp_posts.ID
FROM wp_posts INNER JOIN wp_postmeta ON ( wp_posts.ID = wp_postmeta.post_id )
WHERE 1=1 AND (
wp_postmeta.meta_key = '_ilsi_date'
) AND ((wp_posts.post_type = 'publication' AND (wp_posts.post_status = 'publish' OR wp_posts.post_status = 'acf-disabled')))
GROUP BY wp_posts.ID
ORDER BY wp_postmeta.meta_value DESC
LIMIT 15, 5
[posts] => Array
(
[0] => WP_Post Object
(
[ID] => 38759
[post_author] => 40
[post_date] => 2023-10-05 16:56:40
[post_date_gmt] => 2023-10-05 21:56:40
[post_content] =>
[post_title] => 15 anos da lei de biossegurança: o caso da soja GM no Brasil
[post_excerpt] =>
[post_status] => publish
[comment_status] => closed
[ping_status] => closed
[post_password] =>
[post_name] => 15-anos-da-lei-de-biosseguranca-o-caso-da-soja-gm-no-brasil
[to_ping] =>
[pinged] =>
[post_modified] => 2023-12-08 13:07:17
[post_modified_gmt] => 2023-12-08 18:07:17
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://ilsi.org/publication/15-anos-da-lei-de-biosseguranca-o-caso-da-soja-gm-no-brasil/
[menu_order] => 0
[post_type] => publication
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[1] => WP_Post Object
(
[ID] => 38953
[post_author] => 40
[post_date] => 2023-10-17 11:46:31
[post_date_gmt] => 2023-10-17 15:46:31
[post_content] =>
[post_title] => Bioactive compounds intake in the Brazilian population: Trends and determinants of socioeconomic inequalities between 2008 and 2018
[post_excerpt] =>
[post_status] => publish
[comment_status] => closed
[ping_status] => closed
[post_password] =>
[post_name] => bioactive-compounds-intake-in-the-brazilian-population-trends-and-determinants-of-socioeconomic-inequalities-between-2008-and-2018
[to_ping] =>
[pinged] =>
[post_modified] => 2023-10-17 11:52:52
[post_modified_gmt] => 2023-10-17 15:52:52
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://ilsi.org/publication/bioactive-compounds-intake-in-the-brazilian-population-trends-and-determinants-of-socioeconomic-inequalities-between-2008-and-2018/
[menu_order] => 0
[post_type] => publication
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[2] => WP_Post Object
(
[ID] => 38745
[post_author] => 357
[post_date] => 2023-10-04 03:08:55
[post_date_gmt] => 2023-10-04 07:08:55
[post_content] =>
[post_title] => Effect of Non-Nutritive Sweeteners on Insulin Regulation, Glycemic Response, Appetite and Weight Management: A Systematic Review
[post_excerpt] =>
[post_status] => publish
[comment_status] => closed
[ping_status] => closed
[post_password] =>
[post_name] => effect-of-non-nutritive-sweeteners-on-insulin-regulation-glycemic-response-appetite-and-weight-management-a-systematic-review
[to_ping] =>
[pinged] =>
[post_modified] => 2023-10-04 03:08:55
[post_modified_gmt] => 2023-10-04 07:08:55
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://ilsi.org/publication/effect-of-non-nutritive-sweeteners-on-insulin-regulation-glycemic-response-appetite-and-weight-management-a-systematic-review/
[menu_order] => 0
[post_type] => publication
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[3] => WP_Post Object
(
[ID] => 38131
[post_author] => 351
[post_date] => 2023-09-08 08:55:39
[post_date_gmt] => 2023-09-08 08:55:39
[post_content] =>
Research on gut microbiota has generally focused on fecal samples, representing luminal content of the large intestine. However, nutrient uptake is restricted to the small intestine. Abundant immune cell populations at this anatomical site combined with diminished mucus secretion and looser junctions (partly to allow for more efficient fluid and nutrient absorption) also results in intimate host-microbe interactions despite more rapid transit. It is thus crucial to dissect key differences in both ecology and physiology between small and large intestine to better leverage the immense potential of human gut microbiota imprinting, including probiotic engraftment at biological sensible niches. Here, we provide a detailed review unfolding how the physiological and anatomical differences between the small and large intestine affect gut microbiota composition, function, and plasticity. This information is key to understanding how gut microbiota manipulation, including probiotic administration, may strain-dependently transform host-microbe interactions at defined locations.
This review focusses its narrative on the intimate relationship between the host and its bacterial constituents of the small and large intestine.
Dowload the full paper
or click on the images below to download the EG one-pager summary and graphical abstract

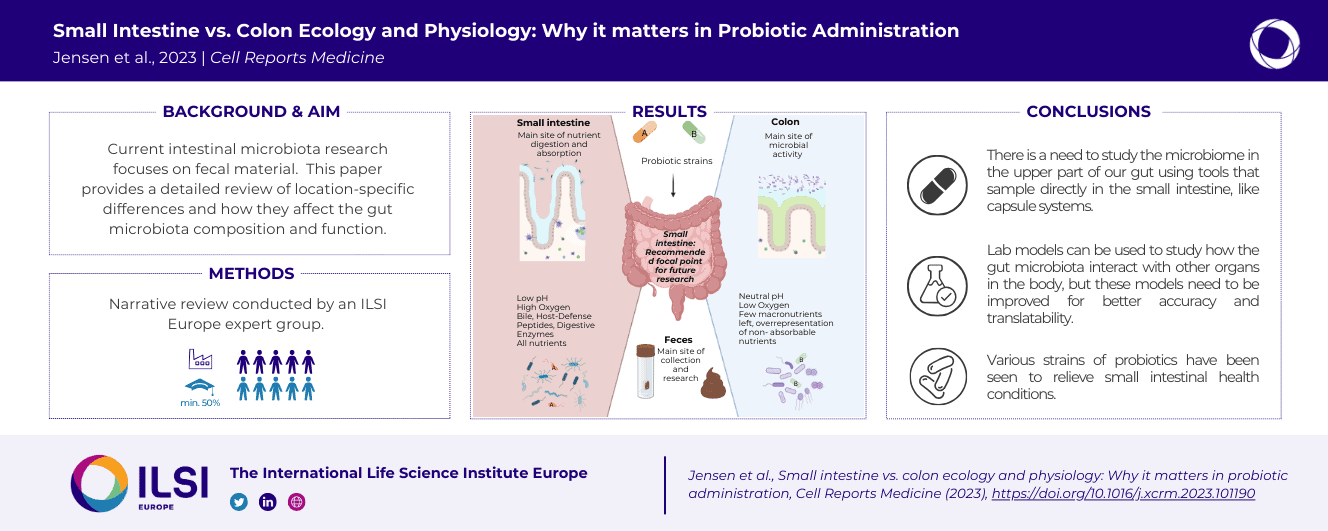
Commissioned by the Probiotics Task Force
[post_title] => Small intestine vs. colon ecology and physiology: Why it matters in probiotic administration
[post_excerpt] =>
[post_status] => publish
[comment_status] => closed
[ping_status] => closed
[post_password] =>
[post_name] => small-intestine-vs-colon-ecology-and-physiology-why-it-matters-in-probiotic-administration
[to_ping] =>
[pinged] =>
[post_modified] => 2023-09-08 08:36:11
[post_modified_gmt] => 2023-09-08 12:36:11
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://ilsi.org/publication/small-intestine-vs-colon-ecology-and-physiology-why-it-matters-in-probiotic-administration/
[menu_order] => 0
[post_type] => publication
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[4] => WP_Post Object
(
[ID] => 38546
[post_author] => 40
[post_date] => 2023-09-26 13:10:37
[post_date_gmt] => 2023-09-26 17:10:37
[post_content] =>
[post_title] => Contribuição do eucalipto geneticamente modificado em uma perspectiva de demanda global e produção sustentável
[post_excerpt] =>
[post_status] => publish
[comment_status] => closed
[ping_status] => closed
[post_password] =>
[post_name] => contribuicao-do-eucalipto-geneticamente-modificado-em-uma-perspectiva-de-demanda-global-e-producao-sustentavel-o-ilsi-atendeu-ao-16o-simposio-isbr-em-st-louis-nos-eua-nas-datas-de-30-de-abril-a-5-d
[to_ping] =>
[pinged] =>
[post_modified] => 2023-09-26 13:12:58
[post_modified_gmt] => 2023-09-26 17:12:58
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://ilsi.org/publication/contribuicao-do-eucalipto-geneticamente-modificado-em-uma-perspectiva-de-demanda-global-e-producao-sustentavel-o-ilsi-atendeu-ao-16o-simposio-isbr-em-st-louis-nos-eua-nas-datas-de-30-de-abril-a-5-d/
[menu_order] => 0
[post_type] => publication
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
)
[post_count] => 5
[current_post] => -1
[before_loop] =>
[in_the_loop] =>
[post] => WP_Post Object
(
[ID] => 38759
[post_author] => 40
[post_date] => 2023-10-05 16:56:40
[post_date_gmt] => 2023-10-05 21:56:40
[post_content] =>
[post_title] => 15 anos da lei de biossegurança: o caso da soja GM no Brasil
[post_excerpt] =>
[post_status] => publish
[comment_status] => closed
[ping_status] => closed
[post_password] =>
[post_name] => 15-anos-da-lei-de-biosseguranca-o-caso-da-soja-gm-no-brasil
[to_ping] =>
[pinged] =>
[post_modified] => 2023-12-08 13:07:17
[post_modified_gmt] => 2023-12-08 18:07:17
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://ilsi.org/publication/15-anos-da-lei-de-biosseguranca-o-caso-da-soja-gm-no-brasil/
[menu_order] => 0
[post_type] => publication
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[comment_count] => 0
[current_comment] => -1
[found_posts] => 791
[max_num_pages] => 159
[max_num_comment_pages] => 0
[is_single] =>
[is_preview] =>
[is_page] =>
[is_archive] =>
[is_date] =>
[is_year] =>
[is_month] =>
[is_day] =>
[is_time] =>
[is_author] =>
[is_category] =>
[is_tag] =>
[is_tax] =>
[is_search] =>
[is_feed] =>
[is_comment_feed] =>
[is_trackback] =>
[is_home] => 1
[is_privacy_policy] =>
[is_404] =>
[is_embed] =>
[is_paged] => 1
[is_admin] =>
[is_attachment] =>
[is_singular] =>
[is_robots] =>
[is_favicon] =>
[is_posts_page] =>
[is_post_type_archive] =>
[query_vars_hash:WP_Query:private] => a55ff190b46cec82b1286c09c4572e35
[query_vars_changed:WP_Query:private] =>
[thumbnails_cached] =>
[allow_query_attachment_by_filename:protected] =>
[stopwords:WP_Query:private] =>
[compat_fields:WP_Query:private] => Array
(
[0] => query_vars_hash
[1] => query_vars_changed
)
[compat_methods:WP_Query:private] => Array
(
[0] => init_query_flags
[1] => parse_tax_query
)
)